
The Atomic Theory, Experimentations, and Its Modern View This is why experimentation is so critical to the scientific process. Based on his own work, he made an assumption that turned out to not be true. This caused him to believe that the formula for water was HO instead of H2O and ammonia was NH instead of NH3.ĭalton had made the same mistake that many had before. He believed that when atoms combined in only one ratio, then it needed to be assumed that it would be a binary ratio. This would help to explain why weight ratios in various gases were simple multiples of each other.ĭalton had another postulate that he included with his initial atomic theory that, unfortunately, made it difficult for the scientific community to accept his ideas in their entirety. If an element reacts, their atoms may sometimes combine into more than one simple whole-number ratio.This indicates that when a chemical reaction occurs, it is because the atoms are being rearranged in such a way that they form a different combination. All chemical reactions are a rearrangement of atoms.A combination may also include more than two. For a compound to form, Dalton suggested with his atomic theory that it would have to be composed of at least two different types of atoms. All compounds are made up of atom combinations.It also means that an element can be identified because its atoms will act like a fingerprint to identify it. This means that any given element has atoms that must be identical in properties, including their mass. All atoms can be identified by mass and properties.This means that everything that is made of matter is composed of atoms, which are indivisible by design. This means that there are five components to the atomic theory that are offered by Dalton. When Dalton proposed his atomic theory, it was based on ideas, assumptions, and principles more than facts that were directly observable. The Four Principles of Dalton’s Atomic Theory Now other items could be added to the salt to change it, but the core atoms of salt are always the same. That means if you were to look at salt crystals, then you would have the same proportions of the base elements, chlorine and salt, no matter how much salt you had or where you got the salt. The law of constant composition says that pure compounds will always have the same proportion of the same elements. It is for this reason that mathematics seeks to create equality and balance. This means if a chemical reaction happens to create something new, then the amount of each element must come from the same starting materials. The law of conservation of mass says that within a closed system, no matter can be created or destroyed. 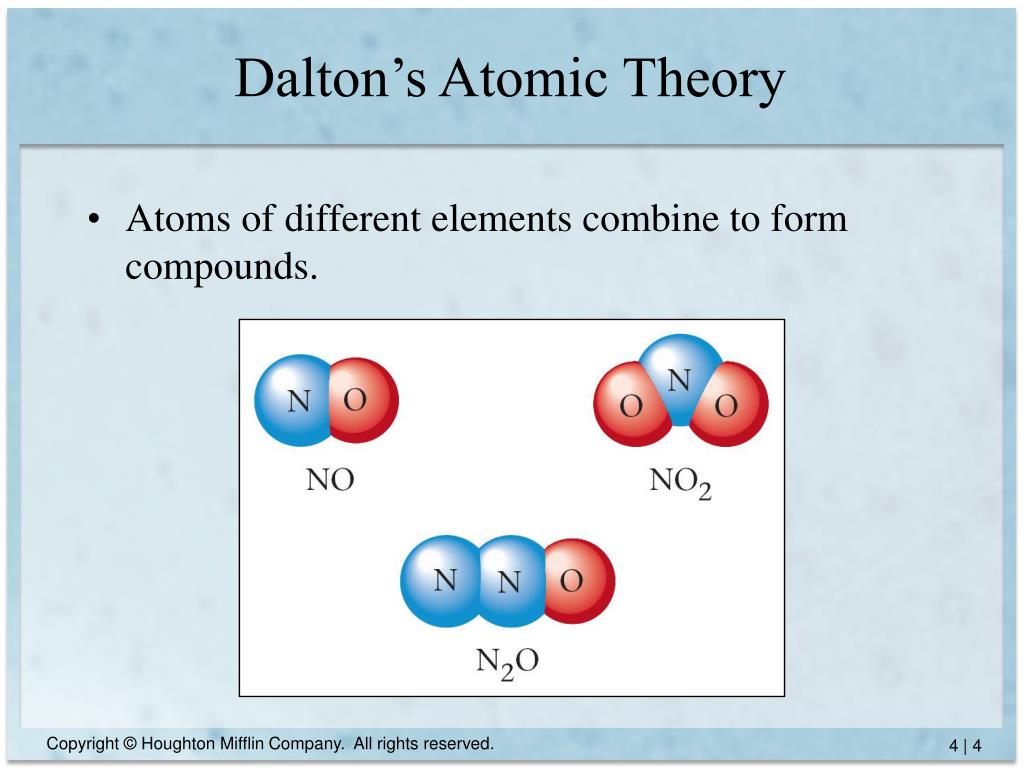
His theory was based on two verified scientific laws: the law of conservation of mass and the law of constant composition. Through different experiments with gases Dalton expanded on this to theorise that atoms vary in size and mass and that compounds had to be made of whole number ratios of atoms.John Dalton’s atomic theory experiment was the first attempt to describe all matter by way of atoms and their properties in a way that was complete. 
This built on the work of Lavoisier and Dalton furthered this proposing that each chemical element is made of atoms of a unique type and they cannot be altered or destroyed but can be combined. He found that 100g of tin will combine with 13.5g or 27g of oxygen and that this could be represented by a 2:1 ratio, for every 2 atoms of oxygen there was one atom of tin. He looked at tin oxide and the combination of masses of oxygen with tin. Dalton used the work of Lavoisier and Joseph Proust to examine the ratios of elements that combine to form compounds and look at their ratio of masses. The work of Lavoisier and the atomists was furthered in the 18th Century by the British scientist John Dalton. It also proved the earlier work of Robert Boyle who hypothesised in 1661 that elements cannot be broken down into simpler substances. This was a crucial breakthrough in the work of atomists in confirming what matter was made of as it was proved that atoms are not created or destroyed when a reaction happens. This led to the theory of the law of conservation of mass. They formulated the key concepts of the law of conservation of mass and the existence of atoms as the building blocks of all matter using their knowledge of chemical reactions.Ī later breakthrough in the discovery of the atomic model came through the work of French chemist Antoine Lavoisier who through a series of experiments found that the total mass of products and reactants in a chemical reactions is always the same. The modern Atomic Model was first developed by two key scientists Lavoisier and Dalton with the help of others.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
